Multifunctional Platforms for Targeted Gene Delivery and Enhanced Intracellular Trafficking
While the transgene expression achieved was relatively low, the work paved the way for the idea of delivering DNA to skeletal muscle for therapeutic purposes. Owing to its relative abundance and continuously high levels of protein expression, skeletal muscle is ideally suited as a target for gene delivery. In addition, skeletal muscle is well vascularized, allowing for the secretion of potential therapeutic proteins for the amelioration of disease. While gene delivery to skeletal muscle holds immense promise, achieving it with great efficiency remains a challenge. Much of the current literature focuses on viral-mediated delivery to skeletal muscle. A renewed interest has emerged in nonviral means of gene delivery. However, even with the nonviral means of gene delivery the major challenges that remain for the successful delivery of nucleic acids to skeletal muscle are (1) cell-specific targeting and (2) the necessary intracellular trafficking to ensure delivery to the nucleus. The development of a DNA delivery platform that features both functional tissue- and cellular compartment-specific targeting will greatly improve delivery of nucleic acids to target tissue. To that end, we are developing a platform will comprise (1) a peptide-functionalized dendrimer for tissue-specific targeting and (2) a bioactive peptide for efficient intracellular trafficking.
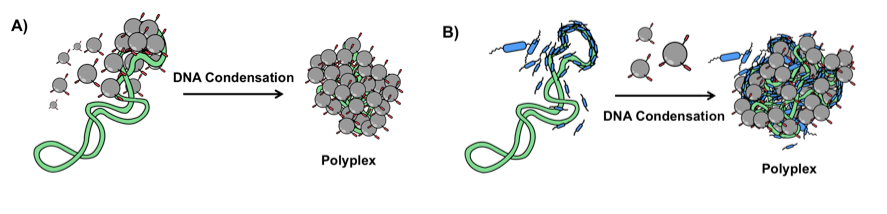
Tissue targeted dendrimer-DNA complex Tissue and nuclear targeted dendrimer-DNA complex

Scanning Electron Micrograph of polyplexes

